Opinion
Patient Access to Biopharmaceutical Innovations: An Industry Perspective, By Anne Sinke, Ph.D., Medical Director Gilead Sciences
On this we can all agree: Everyone deserves access to the best possible medical care and treatments. But the path to care is often shaped by where you live, what you can afford, and how health systems prioritize access. Last year, a Gilead Sciences breast cancer treatment made headlines because it took nearly three years for patients in the Netherlands to gain access, despite the treatment being accessible for some time in most European countries.

Anne Sinke, Ph.D., Medical Director Gilead Sciences Photo by:
The latest reports show that access to innovative medicines in the Netherlands takes on average 459 days and increases by about six weeks each year.[1] In addition, year after year, the same reports show a concerning, declining trend in the proportion of EMA-approved innovations that become accessible to patients in the Netherlands: 31 out of 41 drugs (76%) were accessible between 2016-2019, versus only 24 out of 56 oncology drugs (43%) and 30 out of 66 orphan drugs (45%) between 2020-2023.[2] [3]
This is resulting in troubling and emotionally charged scenarios, with patients in the Netherlands seeking alternative pathways to access treatments in neighboring countries –some paying out of pocket, others turning to crowdfunding, and some even contemplating relocation.
One of the contributing factors to this situation is the Dutch government's 'Lock' ('Sluis') Procedure, introduced in 2015 to regulate the reimbursement of new medicines. Under this procedure, the Dutch National Health Care Institute evaluates a medicine’s efficacy, cost-effectiveness, necessity, and feasibility. If the outcome is positive, it initiates price negotiations, which can be lengthy.
A New Perspective: Balancing Accessibility and Affordability as a Polarity, Not a Problem
Groundbreaking therapies are only valuable if they reach the right patients at the right time and in the right way. But the Netherlands and many other countries are at a crossroads – between the accessibility and affordability of biopharmaceutical innovations. This dilemma is often perceived as a problem, with either a right or wrong solution, thereby causing a heated and polarized debate. Another perspective is to look at this through the lens of the “polarity model.”
The polarity model recognizes the dilemma as a polarity – an issue that is ongoing, unsolvable, and contains seemingly opposing ideas. However, the model also sees the opposing views as two sides of an issue that are both necessary for balance.[4] Solving the polarity requires collaboration between all stakeholders, something that Gilead is strongly in favor of.
In fact, a byproduct of securing patient access to Gilead’s breast cancer treatment in the Netherlands is that it led to our company becoming a more vocal advocate for the role of collaboration and understanding in effecting positive change. We are actively engaged in a dialogue with other stakeholders – including patient advocacy groups, professional medical and scientific societies, healthcare professionals, government health agencies, payers and industry partners – not just in the Netherlands, but worldwide.
An Industry Perspective
Discovering, developing and delivering innovations for life-threatening diseases takes a long time, and involves significant risk and substantial investments. Typically, only one drug emerges from a pool of 10,000 molecules and 10 to 15 years of rigorous research, testing, and regulatory approval, including preclinical studies, clinical trials, and manufacturing optimization.[5] An analysis of the R&D input, output, and outcome of 16 leading research-based pharmaceutical companies over 20 years (2001-2020) showed an average of $2.4 to 6.16 billion total R&D expenditures per new drug.[6]
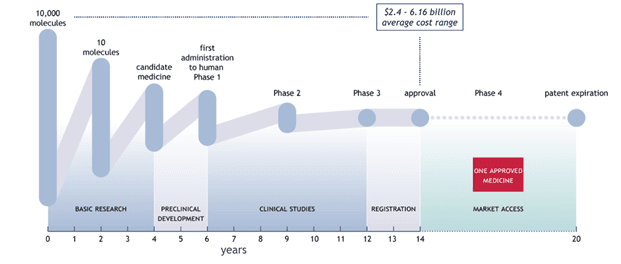
Continuous drug development is associated with significant investments.
Prasad V, Mailankody S. Research and development spending to bring a single cancer drug to market and revenues after approval. JAMA Intern Med 2017;177:1569-75. doi:10.1001/jamainternmed.2017.3601
SiRM, L.E.K. Consulting & RAND Europe, The financial ecosystem of pharmaceutical R&D: An evidence base to inform further dialogue, 2022.
Schuhmacher A, Hinder M, von Stegmann Und Stein A, Hartl D, Gassmann O. Analysis of pharma R&D productivity - a new perspective needed. Drug Discov Today. 2023 Jul 26;28(10):103726. doi: 10.10
At the heart of the issue is the value of innovation. In 1996, people living with HIV were young, not expected to live past 40, and took on average over 20 pills a day. Today, almost half of people living with HIV are over 50, have a normal life expectancy, and take a single daily pill. However, although great progress has been made in the treatment of HIV, new HIV infections are on the rise in many countries and there is still much more innovation needed to end the HIV epidemic for everyone, everywhere.
Similarly, the Dutch Cancer Registry reported recently that the four-year survival rate for cancer patients has risen to 72%, up from 51% thirty years ago clearly showing that great improvements have been made that were attributed to ongoing research into new treatment methods and medications. In addition, survival of patients with newly diagnosed metastatic cancer improved, although modestly and unevenly among the different cancer types, over 30 years.[7] Metastatic cancer remains a very lethal disease.
Therefore, there is still a clear need for better treatment options, better preventive measures and early detection to reduce the incidence of metastatic disease. The challenge is that only 1 out of 20 (5%) of cancer drugs in Phase 1 clinical trials and less than 1 out of 2 (44%) of cancer drugs in Phase 3 clinical trials ever see the light of day.[8]
In the Netherlands, as already stated, this is further exacerbated by the declining accessibility to oncology drugs over time. As the bar graphs below from the EFPIA (European Federation of Pharmaceutical Industries and Associations) Patients W.A.I.T. (Waiting to Access Innovative Therapies) Indicator illustrate, we are moving in the wrong direction.
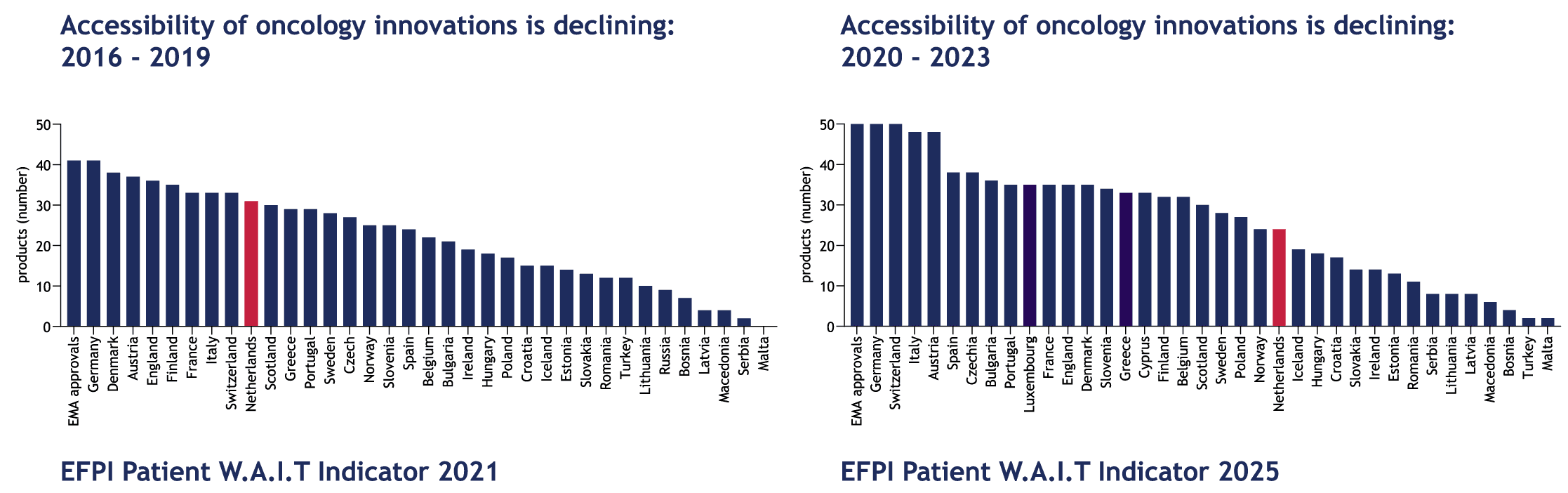
In the Netherlands in particular, oncology drug accessibility is decreasing over time.
Currently, decision-making is driven in part by the perception that spending on innovative medicines is spiraling out of control. The graph below, however, reveals that while medical specialty care spending (blue line) in the Netherlands has steadily increased over six years, the expenditure on add-on expensive medications (red line) has remained relatively stable.[9] However, hospitals do not always recognize these discounts, as the financial benefit often does not return to them."
5 days ago (v0.2)
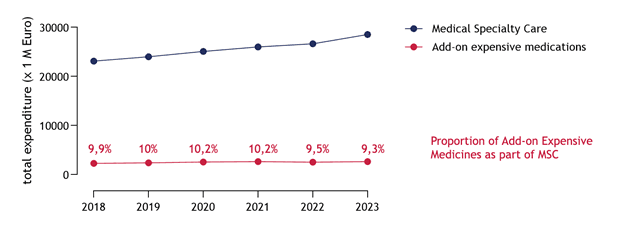
Proportion of add-on innovative medicines as part of Medical Specialty Care (MSC) budget remained stable over time.
NZa Kerncijfers intramurale dure geneesmiddelen 2018 tm 2022 en voorlopige cijfers 2023
Moving Forward, Together
Availability of medicines is shaped by a complex interplay of factors: regulatory approval, government policies, pricing negotiations, willingness to pay, health technology assessments, and healthcare coverage. At the individual level, availability of comprehensive healthcare services, disease awareness, stigma, health literacy, and trust in healthcare institutions play critical roles. Achieving equitable access means aligning political will, economic capacity, and public understanding, in other words, accepting the polarity but working together toward the common goal.
As the accessibility and affordability of pharmaceutical innovations continues to be under pressure, it is of utmost importance that we have an ongoing and constructive dialogue with all relevant stakeholders. We need to understand the medicine’s value, identify a price that reflects its worth, and explore solutions that ensure optimal patient care—both in the Netherlands and beyond. Gilead is open to constructive dialogue and committed to contributing to a workable outcome, always with patients’ best interests at heart.
[1] Patiënten moeten in Nederland nóg langer wachten op nieuwe geneesmiddelen. Vereniging Innovatieve Geneesmiddelen. Published May 13, 2025. https://www.vereniginginnovatievegeneesmiddelen.nl/kennisbank/patienten-moeten-nog-langer-wachten-op-nieuwe-geneesmiddelen/
[2] Newton M, Scott K. EFPIA Patients W.A.I.T. Indicator 2021 Survey. (Troein P, ed.). EFPIA, IQVIA; 2022. Accessed June 19, 2025. https://www.efpia.eu/media/676539/efpia-patient-wait-indicator_update-july-2022_final.pdf
[3] Schwaiger C. Netherlands continues negative trend, sees further decline in drug wait time rankings. Euractiv. Published May 19, 2025. Accessed June 19, 2025. https://www.euractiv.com/section/health-consumers/news/netherlands-continues-negative-trend-sees-further-decline-in-drug-wait-time-rankings/
[4] Leading with Balance: Polarity Management for Effective Leadership. Systems Thinking Alliance. Published November 11, 2024. https://systemsthinkingalliance.org/leading-with-balance-polarity-management-for-effective-leadership/
[5] Research & Development | PhRMA. Phrma.org. Published 2025. https://www.phrma.org/policy-issues/research-development
[6] Schuhmacher A, Hinder M, von Stegmann und Stein A, Hartl D, Gassmann O. Analysis of pharma R&D productivity – a new perspective needed. Drug Discovery Today. 2023;28(10):103726. doi:https://doi.org/10.1016/j.drudis.2023.103726
[7] Luyendijk M, Visser O, Blommestein HM, et al. Changes in survival in de novo metastatic cancer in an era of new medicines. Journal of the National Cancer Institute. 2023;115(6):628-635. doi:https://doi.org/10.1093/jnci/djad020
[8] Thomas D, Chancellor D, Micklus A, et al. Clinical Development Success Rates and Contributing Factors 2011-2020.; 2021. https://go.bio.org/rs/490-EHZ-999/images/ClinicalDevelopmentSuccessRates2011_2020.pdf
[9] NZa Kerncijfers intramurale dure geneesmiddelen 2018 tm 2022 en voorlopige cijfers 2023 - Nederlandse Zorgautoriteit. Overheid.nl. Published 2018. Accessed June 19, 2025. https://puc.overheid.nl/nza/doc/PUC_775444_22/




